Polar acetone 5 Liter pletfjerner

Polar acetone 5L
Category: Science & Tech Also called: 2-propanone Or: dimethyl ketone Related Topics: ketone cordite On the Web: Australian Government - Department of Climate Change, Energy, the Environment and Water - Acetone (Dec. 29, 2023) (Show more) See all related content →

Acetone Formula Properties, Preparation & More Embibe
Acetone is a polar molecule since it possesses a polar bond and the dipole is not cancelled by the molecular structure. Test Your Knowledge On Polarity! Q 5. Put your understanding of this concept to test by answering a few MCQs. Click 'Start Quiz' to begin!
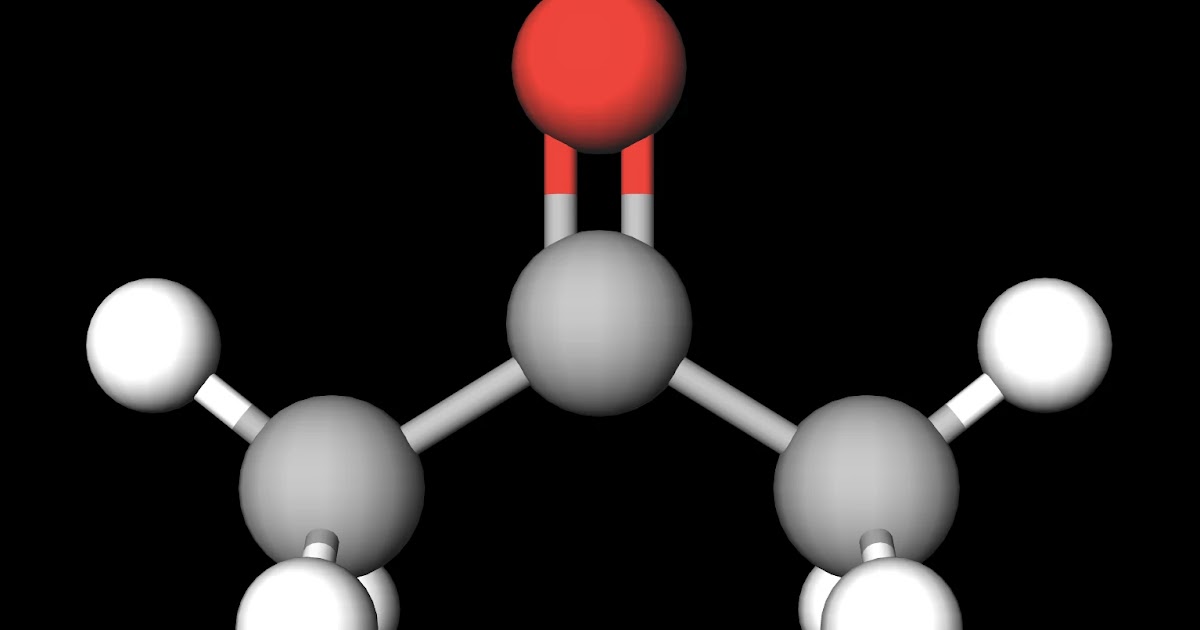
MakeTheBrainHappy Is Acetone Polar or Nonpolar?
Water Acetic Acid Ethylene Glycol Methanol Ethanol Isopropanol Pyridine Acetonitrile Nitromethane Diethylamine Aniline Dimethylsulfoxide Ethyl Acetate Dioxane Acetone Dicholoroethane Tetrahydrofuran Dicholoromethane Chloroform Diethyl Ether Benzene Toluene Xylene Carbontetrachloride Cyclohexane Petroleum ether Hex.

Acetone Uses in Daily Life StudiousGuy
Acetone molecules are polar because of the positive and negative charges formed by their carbonyl group. The molecules that compose acetone do have nonpolar covalent bonds within their overall structure, such as their carbon to hydrogen and carbon to carbon bonds.

Is C3H6O Polar or NonPolar? (Acetone) YouTube
0.447 g/100 mL. 4.7 mL (gas)/ 100 mL. 6.5 mL (gas)/ 100 mL. 8.0 g/100 mL. This table shows that alcohols (in red) have higher boiling points and greater solubility in H haloalkanes and alkanes with the same number of carbons. It also shows that the boiling point of alcohols increase with the number of carbon atoms.

Acetone Solvent Able Westchem
So acetone is a polar aprotic solvent. Some other polar aprotic solvents are listed below. Answer link. Acetone is a polar aprotic solvent. A solvent is polar if it has a dipole moment greater than 1.6 D and a dielectric constant greater than 5. The values for acetone are µ = 2.88 D and ε = 21. So acetone is a polar solvent.
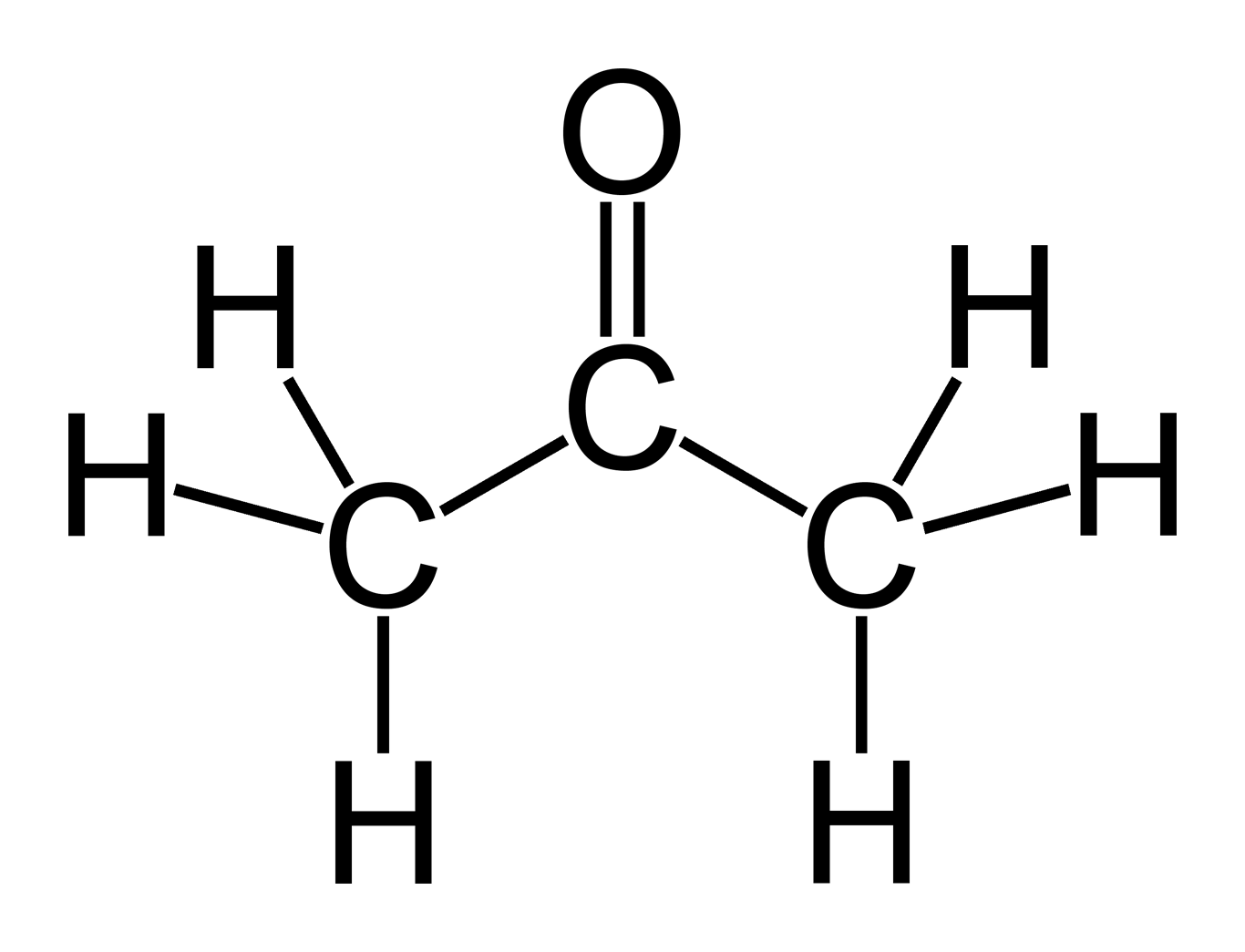
Difference Between Acetone vs. NonAcetone Difference Between
μ = δd μ = δ d. The bond dipole is modeled as +δ — δ- with a distance d between the partial charges +δ and δ-. It is a vector, parallel to the bond axis, pointing from minus to plus, as is conventional for electric dipole moment vectors. In your case, acetone has a higher dipole moment than ethanol because it has a carbonyl group more.

Acetone Physical Properties of Acetone
Acetone contains a polar C=O double bond oriented at about 120° to two methyl groups with nonpolar C-H bonds. The C-O bond dipole therefore corresponds to the molecular dipole, which should result in both a rather large dipole moment and a high boiling point. Thus we predict the following order of boiling points:

Is Acetone Polar Nonpolar Or Ionic girtbir Coub
Acetone is a small molecule that has very non-polar and polar properties simultaneously. Its polar C=O bond makes it miscible (soluble) in water , while its non-polar methyl (CH 3 ) groups can interact with non-polar compounds.
Polar Polar acetone 2,5 L DMT Værktøj
Acetone is a polar solvent, but has non-polar properties. It can remove, dissolve, and clean non-polar substances and stains, such as wax and tree sap, due to its 2 non-polar bonds.

Polar acetone 1 Liter Pletfjerner
However, acetone is still considered a polar aprotic solvent, despite the fact that it is relatively acidic, and not significantly less acidic than alcohols. Then again, acetone (and other carbonyl containing solvents) are, indeed, poor solvents when using strong bases due to their relatively high acidity.

Is acetone polar or nonpolar? Will NaCl dissolve in it? YouTube
Take acetone (C 3 H 6 O) for example. Acetone is polar because there is a partial negative charge on the oxygen and a partial positive charge on the rest of the molecule. This is an aprotic solvent because the highly electronegative atom, oxygen, is not bonded to a hydrogen atom. The hydrogen atoms are bonded to the carbon instead, which is a.

Why Is Acetone Considered A Polar Aprotic Solvent valodil 💣 Coub
To determine if C3H6O (Acetone) is a polar or non-polar molecule we need to look at the Lewis structure, molecular geometry, and the electronegativity of the atoms in C3H6O..more.more.
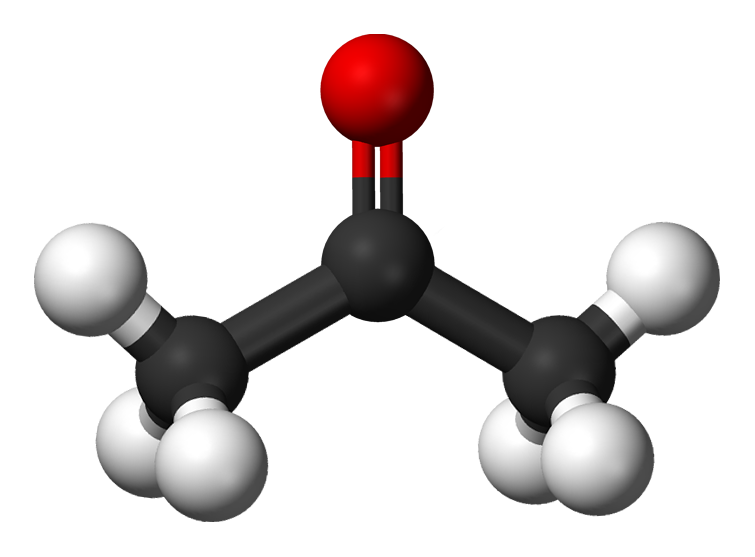
Chemistry Molecule Acetone
Despite this, it is nonpolar. Why? The dielectric constant of chloroform is 4.81 F/m which, being a small value makes it nonpolar. What are Polar Solvents? Polar solvents are the solvents that have a polar bond, the basis of which is the electronegativity difference between constituent atoms.

Is acetone a polar or non polar solvent? YouTube
Is acetone polar or nonpolar? Because of the carbonyl group, acetone is a somewhat polar molecule. There are different degrees of polarity, and acetone is less polar than water, because only part of the acetone molecule has a polar bond. More examples of polar molecules.

Polar acetone 5 Liter pletfjerner
Acetone (C3H6O) is a POLAR molecule because the Oxygen (O) present in the molecule is more electronegative, which causes the partial positive (ẟ+) and partial negative (ẟ-) charge to appear on the molecule. These ẟ+ and ẟ- charges are responsible to make the entire acetone molecule polar.