Simple Method for writing Lewis Structures of the phosphate ion(PO4)3

Structure of PO43 ion Phosphate ion structure Lewis dot method
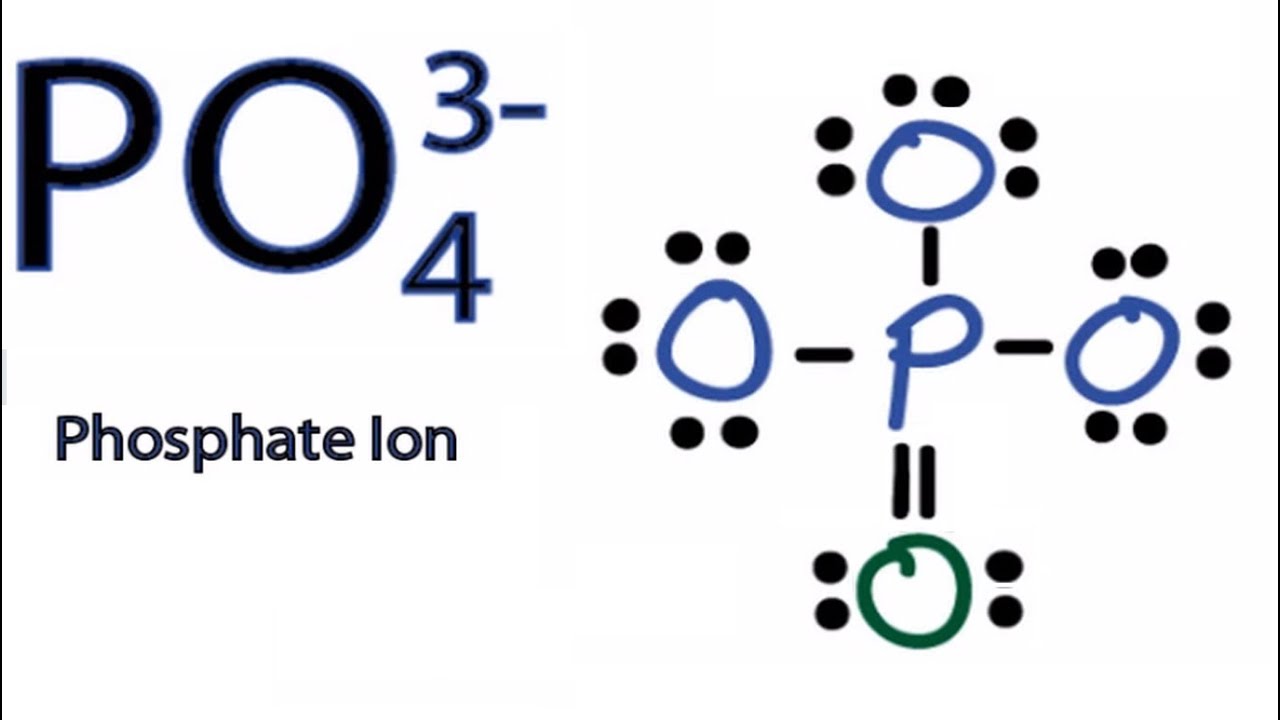
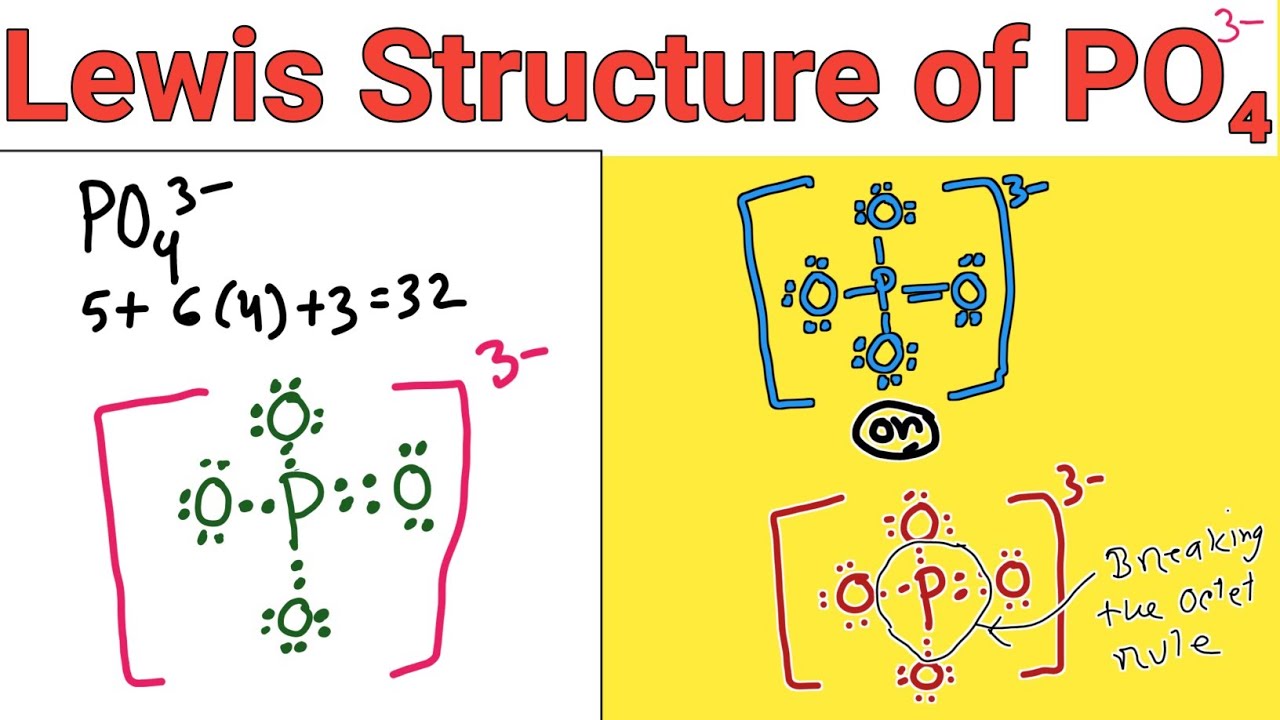
Drawing the Lewis Structure for PO 4 3-Viewing Notes: PO 4 3-has a charge of -3 (that makes it a negative ion or anion). That means that it has an three extra electrons that needs to be taken into account.. Let's do the Lewis structure for PO4 3-. Phosphorus has 5 valence electrons. Oxygen has 6, we've got 4 Oxygens. This negative 3 up here.
PO4 3 Lewis Structure How to Draw the Lewis Structure for PO43 YouTube
Steps of drawing PO4 3- lewis structure Step 1: Find the total valence electrons in PO4 3- ion. In order to find the total valence electrons in PO4 3- ion (phosphate ion), first of all you should know the valence electrons present in phosphorus atom as well as oxygen atom. (Valence electrons are the electrons that are present in the outermost orbit of any atom.)

Lewis and Resonance Structures of The Phosphate Ion (PO43) YouTube
In this video, Let us discuss how to write Lewis structure of PO43-, Phosphate ion easily. A simple notation used to represent valence electrons in an atom is called Lewis symbol. According.

What Is The Formal Charge On Phosphorus In A Lewis Structure Drawing Easy
The Lewis structure for PO43- is shown.

Lewis Dot Structure of Phosphate (PO4 3).....No More Confusion
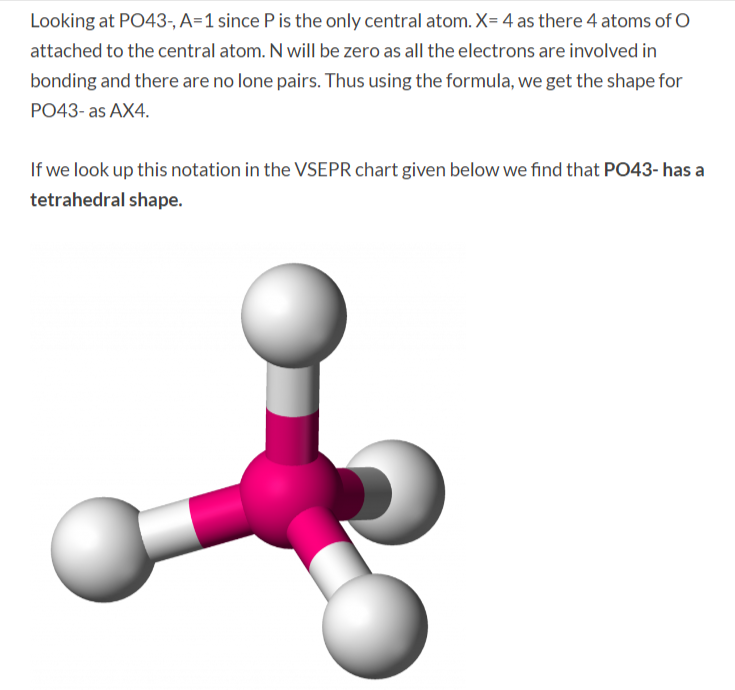
Geometry. PO43- Geometry and Hybridization. Phosphorous is the central and there are 5 + 4×6 + 3 = 32 electrons. Give each oxygen three lone pairs: All the electrons are used and the only thing to fix is the ionic charge. As drawn, it would be a -4, therefore, we give one lone pair from an oxygen to make a double bond with the P which can.
PO43 Lewis Structure (Phosphate Ion) YouTube
Hey Guys!Did you know that Phosphorus can have expanded orbitals and can accommodate more than 8 electrons in its outer shell? Well, such information helps t.
PO43LewisStructureHybridizationPolarityandMolecularGeometry
Lewis structure of phosphate ion is drawn clearly in this tutorial step by step. Total valence electrons concept is used to draw the lewis structure of PO 43- ion. In lewis structure, there should be charges on atoms. Phosphate ion | PO 43- Phosphate ion is one of the oxyanion of phosphorous. Phosphorous is at +5 oxidation state in PO 43-.
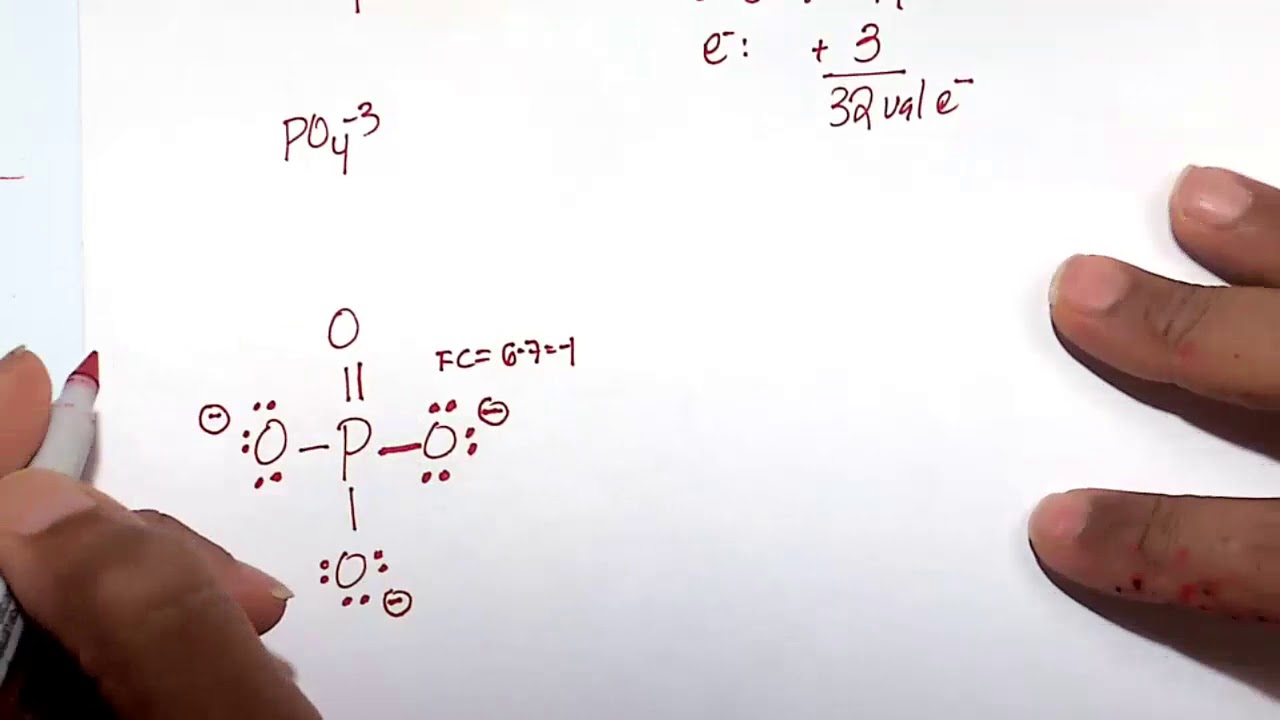
Resonance The resonance structure of the phosphate ion (PO4(3)) YouTube
PROBLEM 4.2.4 4.2. 4. Methanol, H 3 COH, is used as the fuel in some race cars. Ethanol, C 2 H 5 OH, is used extensively as motor fuel in Brazil. Both methanol and ethanol produce CO 2 and H 2 O when they burn. Write the chemical equations for these combustion reactions using Lewis structures instead of chemical formulas.

PO43 lewis structure, molecular geometry, hybridization, and bond angle
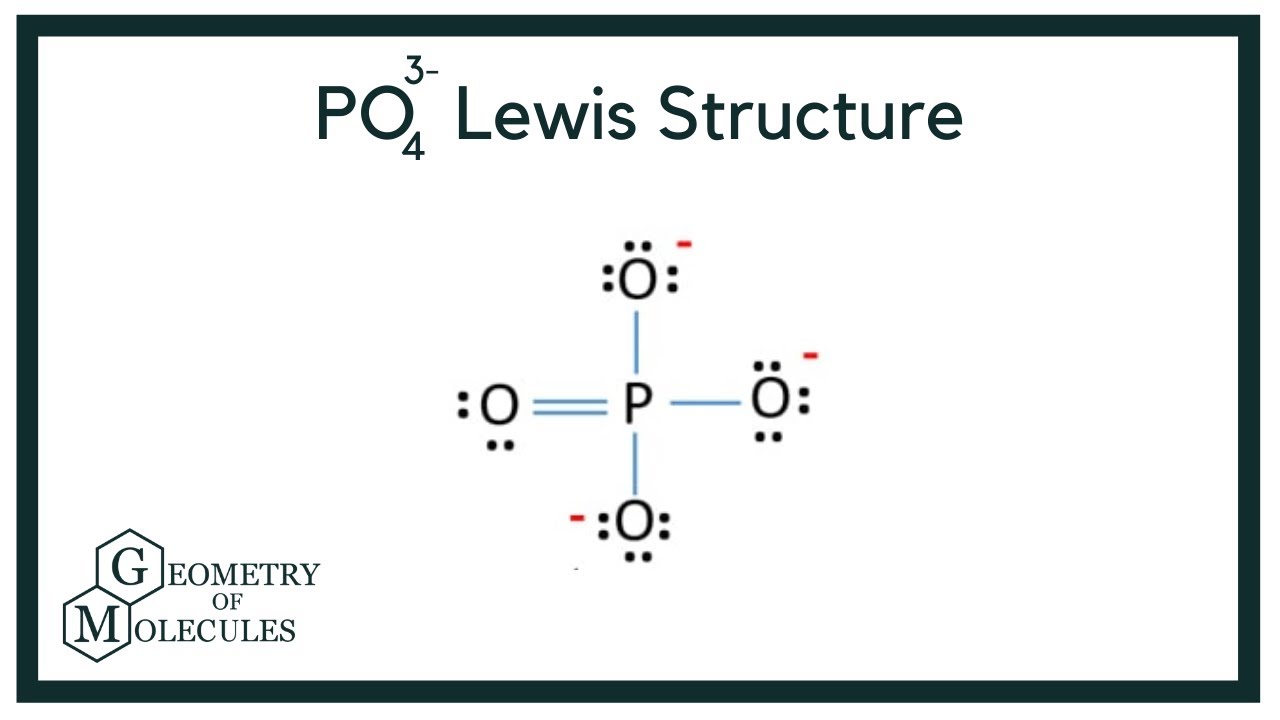
The PO4 3- ion has a Lewis structure with a central phosphorus atom bonded to four oxygen atoms. The phosphorus atom has a formal charge of +3, while each oxygen atom has a formal charge of -1. The Lewis structure of PO4 3- shows that it has a tetrahedral molecular geometry.
Lewis structure of PO4 3 (Phosphate ion) YouTube
PO4 3- is a chemical formula for Floroform. It consists of one sulphur atom and four oxygen atoms.

PPT Bonding General Concept PowerPoint Presentation, free download
How to draw lewis structure of PO43-? The Lewis structure of a phosphate [PO4]3- ion consists of one phosphorus (P) atom and four atoms of oxygen (O). The phosphorus atom is present at the center while the oxygen atoms occupy terminal positions. There are a total of 4 electron density regions around the central phosphorus atom in [PO4]3-.
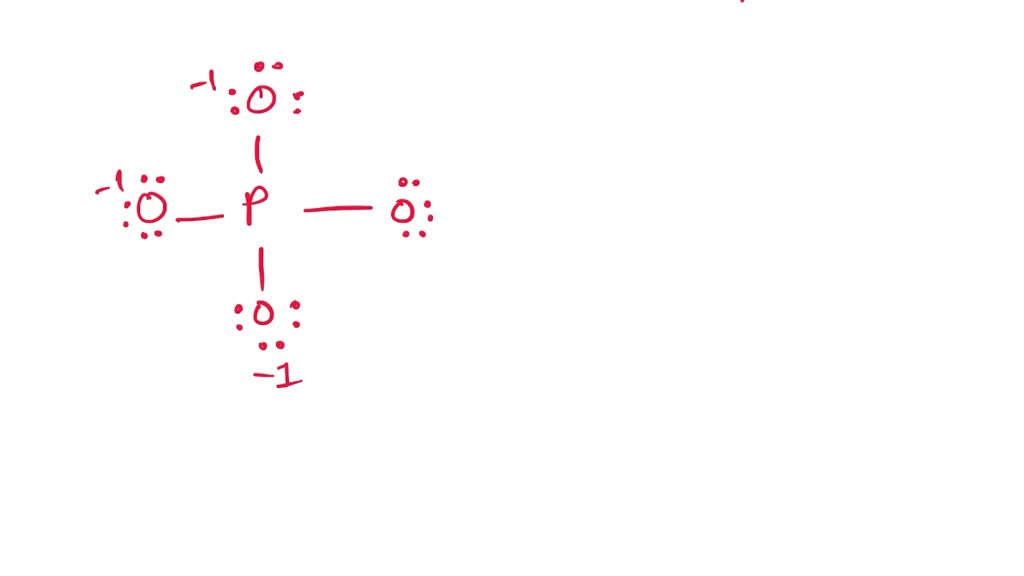
SOLVED The formal charge on the phosphorua atom in resonance structure
In the PO 43- Lewis structure, there is one double bond and three single bonds around the phosphorus atom, with four oxygen atoms attached to it. One oxygen atom with a double bond has two lone pairs, and three oxygen atoms with single bonds have three lone pairs. Also, there is a negative (-1) charge on the three oxygen atoms with single bonds.

In PO43 , the formal charge on each oxygen atom and the P O bond order
In the Lewis structure of PO43- there are a total of 32 valence electrons. For the Lewis structure for PO4 3- you should take formal charges into account to find the best Lewis structure for the molecule. Remember, PO4 3- has a negative three charge on the molecule. For the Lewis structure you'll need to have a total charge for the molecule of 3-.

The Lewis structure for PO43 is shown. Chemistry
A step-by-step explanation of how to draw the PO43- Lewis Dot Structure (Phosphate ion).For the PO4 3- structure use the periodic table to find the total num.

Lewis Structure, Hybridization, Polarization, and Molecular Geometry of
PO43- Molecular Geometry / Shape and Bond Angles Wayne Breslyn 726K subscribers Join Subscribe Subscribed 187 Save 56K views 10 years ago A quick explanation of the molecular geometry of PO43-.

Resonance Structures for PO4 3 (Phosphate ion) YouTube
Phosphate is a very weak oxidizing agent. Since the phosphorus is in its highest oxidation state in phosphate ion, this ion cannot act as a reducing agent. This page titled Phosphate Ion (PO₄³⁻) is shared under a CC BY-NC-SA 4.0 license and was authored, remixed, and/or curated by James P. Birk. Phosphate ion is a reasonably strong base.